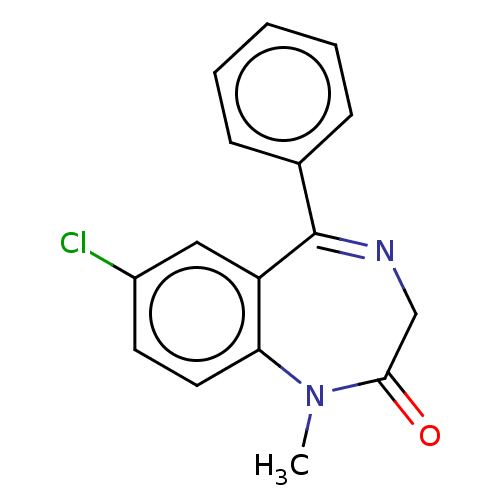
 BDBM50000766 CHEMBL12 DIAZEPAM US9271961, Diazepam
BDBM50000766 CHEMBL12 DIAZEPAM US9271961, Diazepam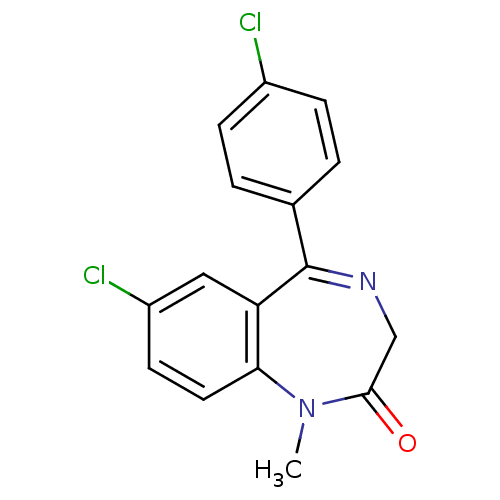 Ro5-4864 4 -chlorodiazepam 7-chloro-5-(4-chlorophenyl)-1-methyl-2,3-dihydro-1H-1,4-benzodiazepin-2-one BDBM22040 4' Cl-diazepam CHEMBL286346 Ro 5-4864 7-chloro-5-(4-chlorophenyl)-1-methyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one Ro-4864 Ro-05-4864
Ro5-4864 4 -chlorodiazepam 7-chloro-5-(4-chlorophenyl)-1-methyl-2,3-dihydro-1H-1,4-benzodiazepin-2-one BDBM22040 4' Cl-diazepam CHEMBL286346 Ro 5-4864 7-chloro-5-(4-chlorophenyl)-1-methyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one Ro-4864 Ro-05-4864
- Wieland, HA; Lüddens, H Four amino acid exchanges convert a diazepam-insensitive, inverse agonist-preferring GABAA receptor into a diazepam-preferring GABAA receptor. J Med Chem 37: 4576-80 (1995)
- Gunnersen, D; Kaufman, CM; Skolnick, P Pharmacological properties of recombinant "diazepam-insensitive" GABAA receptors. Neuropharmacology 35: 1307-14 (1996)
- Braestrup, C; Squires, RF Specific benzodiazepine receptors in rat brain characterized by high-affinity (3H)diazepam binding. Proc Natl Acad Sci U S A 74: 3805-9 (1977)
- Yang, W; Drewe, JA; Lan, NC Cloning and characterization of the human GABAA receptor alpha 4 subunit: identification of a unique diazepam-insensitive binding site. Eur J Pharmacol 291: 319-25 (1995)
- ChEBML_178942 Anxiolytic activity by displacement of [3H]diazepam from GABA-A receptor in rat synaptosomal membranes
- ChEMBL_774144 (CHEMBL1908239) Mechanism based inhibition of dog cytochrome P450 CYP2B11 measured by Diazepam N1-demethylation using liver microsomes
- ChEMBL_774145 (CHEMBL1908240) Mechanism based inhibition of dog cytochrome P450 CYP2B11 measured by Diazepam N1-demethylation using recombinant enzyme
- Biological Assay Example 8 Using a procedure similar to that described in Biological Test Example 6, the inhibition of cytochrome P450 2C19-isoenzyme catalysed N-demethylation of [N-methyl-14C]-Diazepam by the test compound was assayed at 37 � C. with human recombinant cytochrome P450 2C19.
- Biological Assay Example 8 Using a procedure similar to that described in Biological Test Example 6, the inhibition of cytochrome P450 2C19-isoenzyme catalysed N-demethylation of [N-methyl-14C]-Diazepam by the test compound was assayed at 37° C. with human recombinant cytochrome P450 2C19.
- Radioligand Binding Assay Radioligand binding assays were carried out in a volume of 200 mL (96-well plates) which contained 100 mL of cell memebranes, [3H]flumazenil at a concentration of 1 nM for α1, α2, α3 subunits and 0.5 nM for α5 subunits and the test compound in the range of 10-10−3x10−6 M. Nonspecific binding was defined by 10−5 M diazepam and typically represented less than 5% of the total binding. Assays were incubated to equilibrium for 1 hour at 4 C. and harvested onto GF/C uni-filters (Packard) by filtration using a Packard harvester and washing with ice-cold wash buffer (50 mM Tris; pH 7.5). After drying, filter-retained radioactivity was detected by liquid scintillation counting.
- Radioligand Binding Assay Radioligand binding assays were carried out in a volume of 200 mL (96-well plates) which contained 100 mL of cell membranes, [3H]flumazenil at a concentration of 1 nM for α1, α2, α3 subunits and 0.5 nM for α5 subunits and the test compound in the range of 10-10−3×10−6 M. Nonspecific binding was defined by 10−5 M diazepam and typically represented less than 5% of the total binding. Assays were incubated to equilibrium for 1 hour at 4° C. and harvested onto GF/C uni-filters (Packard) by filtration using a Packard harvester and washing with ice-cold wash buffer (50 mM Tris; pH 7.5). After drying, filter-retained radioactivity was detected by liquid scintillation counting.
- Binding Assay Receptor binding assays were performed in 96-well format in deep-well plates. For each 96-well plate one ampulle of membrane homogenate was thawed and diluted in binding buffer (50 mM Tris pH=7.4, 100 mM KCl) and 200 μL was dispensed into each well. Radioligand [3H]Rol51788 (Perkin Elmer: NET757250UC) was prepared in binding buffer and added to each well in 50 μL volume to give final concentration of 0.5 nM. Test compounds in suitable concentration(s) were added in additional 50 μL. The final assay volume was 300 μL. Incubation was carried out for 60 minutes at 4?? C. For non-specific binding 10 μM unlabeled diazepam was used. After incubation samples were filtered over UniFilter*GF/B using Filtermate Harvester (Perkin Elmer) and washed with 5??1 mL binding buffer. The plate was dried at 40?? C. for an hour and 40 μL Microscint (Perkin Elmer) scintillation cocktail was added to each well. The plate was read in Microbeta (Perkin Elmer).
- Binding Assay for γ2-Containing GABAA Subtypes Table 1: Radioligand binding assays were carried out in a volume of 200 μL (96-well plates) which contained 100 μL of cell membranes, [3H]Flumazenil at a concentration of 1 nM and the test compound in the range of 0.1�10−9 to 30�10−6 M. Nonspecific binding was defined by 10−5 M Diazepam and typically represented less than 5% of the total binding. Assays were incubated to equilibrium for 1 hour at 4� C. and harvested onto GF/C uni-filters (Packard) by filtration using a Packard harvester and washing with ice-cold wash buffer (50 mM Tris; pH 7.5). After drying, filter-retained radioactivity was detected by liquid scintillation counting. Ki values were calculated using Exel-Fit (Microsoft) and are the means of two determinations. The compounds of the accompanying examples were tested in the above described assay, and the preferred compounds were found to possess large K, value for displacement of [3H]Flumazenil from the α1β3γ2 subtype of the human GABAA receptor of 100 nM or above. Most preferred are compounds with a Ki α1β3γ2 (nM)>300.

